Research Misconduct
At Hackensack Meridian Health, we are dedicated to the highest standards of integrity in all our research. Upholding these standards is essential for advancing medical science, ensuring patient safety, and maintaining public trust.
We believe that every member of our network shares the responsibility to ensure that our research is conducted ethically and responsibly. Hackensack Meridian Health is committed to addressing any concerns about research misconduct promptly, thoroughly, and fairly.
HMH’s Research Compliance division is responsible for overseeing and administering HMH’s Policy on Research Misconduct. The Research Compliance Officer is the designated Research Integrity Officer (RIO) and is a resource for anyone with concerns or questions about possible research misconduct.
HMH defines research misconduct, in accordance with federal regulations, as any fabrication, falsification, or plagiarism in the proposing, performing or reviewing of research or reporting of research results. Research misconduct does not include honest error or differences of opinion.
|
Type of Research Misconduct |
Definition |
|
Fabrication |
The making up of data or results and the recording or reporting thereof. |
|
Falsification |
The manipulation of Research materials, equipment or processes, or the change or omission of data or results such that the Research is not accurately represented in the Research Record. |
|
Plagiarism |
The appropriation of another person’s ideas, processes, results or words without giving appropriate credit. |
If You Have a Concern
- Discuss with a trusted chair, supervisor or other official
- Contact the Research Compliance Officer, Dr. Michelle Benson
- Anonymous reporting via HMH’s ComplyLine
Retaliation of any kind, against those who report a concern in good faith is not tolerated by HMH.
These are the main stages following an allegation of research misconduct. Each case is unique and not every case may follow each of these stages.
- Allegation received. An initial review (assessment) is conducted to ensure the allegation is sufficiently credible and meets the definition of research misconduct
- Inquiry. A review of the allegation to determine if an investigation is warranted.
- Investigation. A review of the factual record resulting in either a finding of misconduct or dismissal.
- Adjudication. Finding of Research Misconduct accepted, rejected or modified by Research Institutional Official (Chief Research Officer)
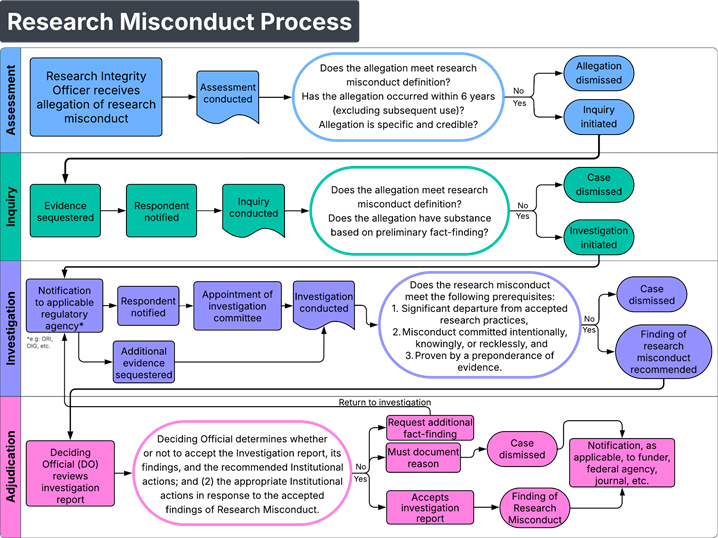
A finding of research misconduct must meet the following prerequisites:
- Significant departure from accepted practices of the relevant research community, and
- Misconduct committed intentionally, knowingly, or recklessly, and
- Proven by a preponderance of evidence.
We take every report about research conduct seriously and to ensure a fair process confidentiality is paramount for all those involved in research misconduct process. Information is only shared with people who absolutely need it for the review, or in rare cases when the law requires us to report to a government office.
In addition, all actual or perceived claims of retaliation are pursued to the fullest extent possible by the Research Misconduct policy and institutional whistleblower protections.
- Clinical Research Billing Compliance
- Conflicts of Interest
- Data Management and Sharing
- Export Controls
- Foreign Interactions and Research Security
- Privacy and Cyber Security Considerations for Research
- Research Compliance
- Research Misconduct
- Research Compliance Audit and Monitoring Program
- Responsible Conduct of Research
